Recycling of electrochemical storage devices (ESDs) or energy devices may involve recovering valuable materials from end-of-life batteries or production rejects.
In March 2022, Accurec Recycling GmbH was granted European patent EP3836290B1 designating the UK, titled Method for Decomposition of Electrochemical Storage Devices and Thermal Treatment Device.
The patented method comprises a first process step (P1) for the decomposition of ESDs and removal of electrolytes and reactive substances, via a thermal treatment performed in a process chamber with a reducing atmosphere.
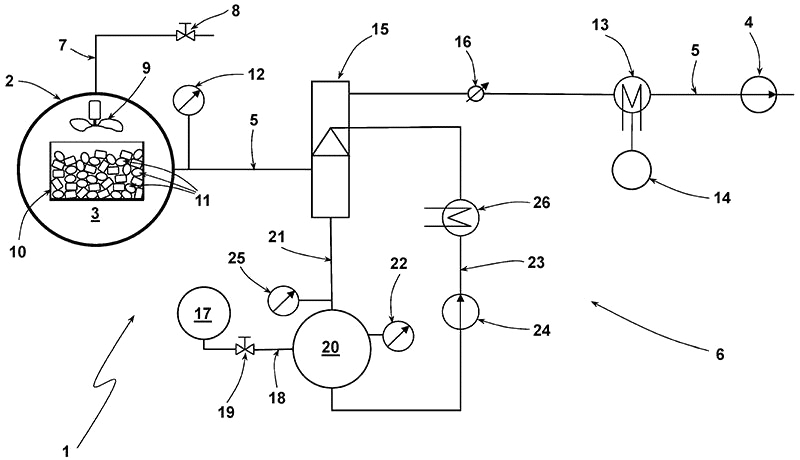
In the illustrated example, the thermal treatment system (1) comprises a vacuum furnace (2), a process chamber (3) and a vacuum pump (4). The pump is used for evacuating the chamber (3) and is connected to the furnace via the suction line (5).
The chamber may be configured to receive a container (10) of a batch of ESDs (11).
A pressure measurement sensor (12) may be arranged in fluid communication with the chamber.
Process step 1 may commence with evacuation of the chamber to a negative pressure relative to the ambient pressure. The chamber may be evacuated to a pressure of less than 10mbar.
The ESDs in the chamber may be heated to a first temperature, causing the electrolytes in the ESDs to evaporate. The electrolytes may include, e.g., ethylene carbonate, dimethyl carbonate and ethyl methyl carbonate.
An increase in the vapour pressure by evaporation of the electrolytes may open the ESDs. A temperature hold may be employed to ensure that all the ESDs are opened.
Process exhaust gases containing the evaporated electrolytes in the vapour phase may be produced within the chamber. These gases may be drawn out of the chamber, e.g., via suctioning. A vacuum condenser (13) may be used to recover the electrolytes into an electrolyte tank (14).
The thermally treated material is then subjected to a second process step (P2), for the separation of secondary raw materials. The ESDs are heated to a second temperature, before the process chamber is vented and cooled allowing removal of the decomposed ESDs.
At the start of P2, the pump may be switched off or the pressure control valve closed. The resulting pressure build-up in the chamber is monitored and controlled to avoid exceeding a certain level, e.g., 600-700mbar.
The process gases may form a reducing atmosphere, while circulating the chamber via convention, promoting uniform heat distribution. Optionally, a fan (9) may be used for circulating the process gases.
The reducing atmosphere may result in a relatively low proportion of metal oxides in the pyrolytically decomposed material as compared with other methods. The majority of the non-oxide metal compounds may be easily separated via post-processing methods e.g., via a magnetic separator.
As a safety measure, an inert gas may be controllably supplied into the chamber via supply line (7) and value (8).
The system may further include an exhaust gas processing system (6). A pressure control value (16) may be connected downstream of this system. At the start of P2, the vacuum condenser may be deactivated, and an injection condenser (15) may be activated – for removing off-gases, such as hydrofluoric acid.
A condensate tank (20) having a level detector (22) may receive condensates from the injection condenser via condensate line (21). The condensate tank may feed back to the injection condenser via line (23) having a pump (24) and heat exchanger (26).
A pH meter (25) may be used to record the pH value of the condensate from the injection condenser. Lye, e.g., caustic soda, from a suds tank (17) connected to the condensate tank via line (18) and having a valve (19), may be introduced into the collection tank when required to neutralise the acid.
Read the full patent here.
This article first appeared in the September 2022 issue of Materials World, the member magazine of the Institute of Materials, Minerals and Mining.
The Patent Eye blog offers a glimpse into the future by showcasing newly granted patents. Join us as we explore the latest innovations shaping our world.
The Patent Eye blog is made available by Alphabet Intellectual Property for informational purposes, in particular, sharing of newly patented technology from granted and published patents available in the public domain. It is not meant to convey legal position on behalf of any client, nor is it intended to convey specific legal advice.
Craving intellectual stimulus? Peer into the insights brought to you by The Patent Eye and explore our latest articles.